Toxicology and bioinformatic studies
The development and use of technological additives require careful assessment of their potential toxicity on human health and their eco-toxicity on the environment. In the case of nanomaterials, microplastics (MP) and nanoplastics (NP), methods tailored to the characteristics of these products must be applied, including the choice of in vitro methods and appropriate bioassays that mirror possible exposure pathways.
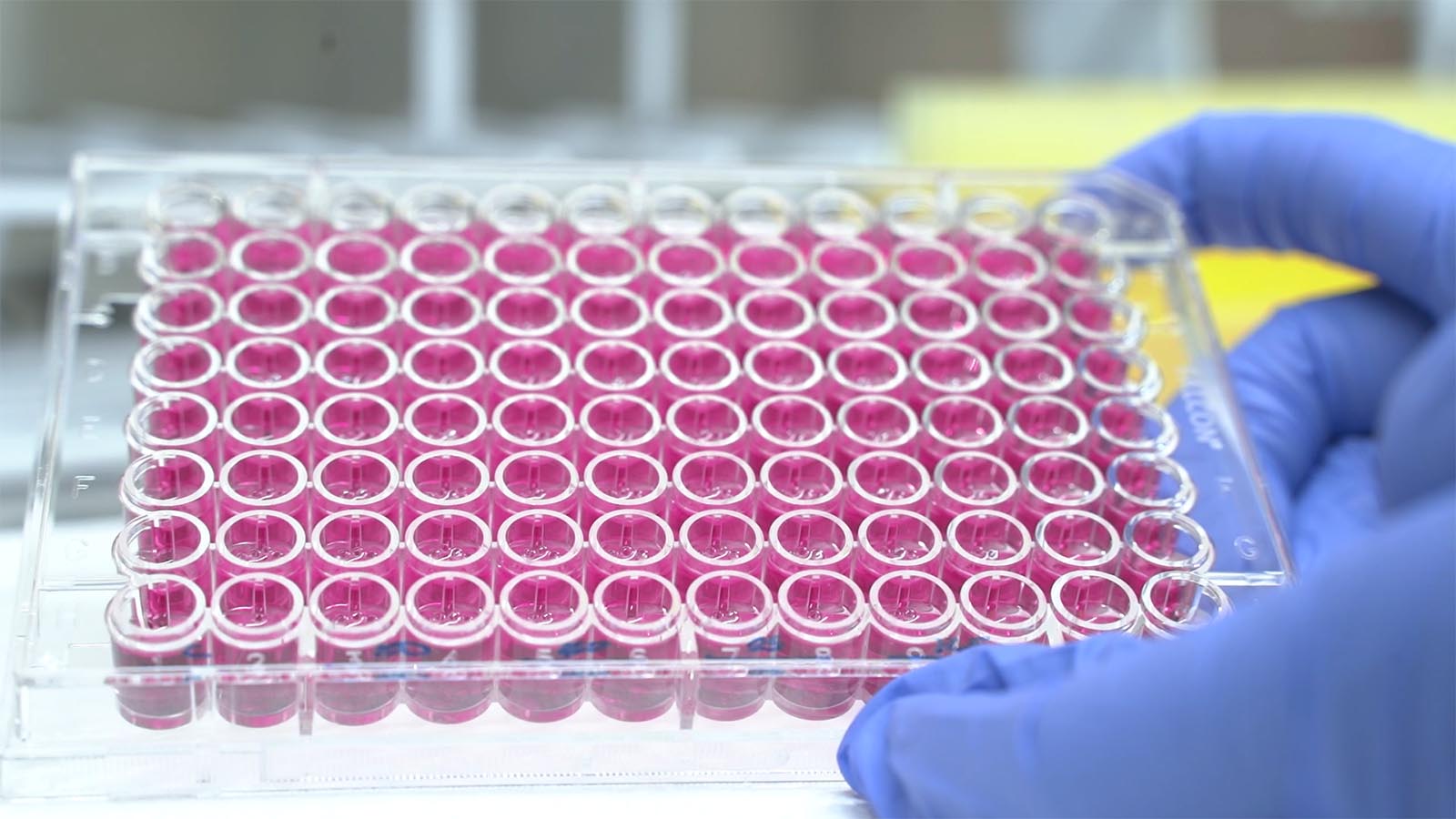
At ITENE we analyse the toxicity of nanomaterials and micro and nanoplastics, as well as that of additives and ingredients used in key areas such as packaging, cosmetics, toiletries, biocides and plant protection products. These tests can be carried out either using computational methods (in silico), which enable us to predict any potentially harmful effects, or experimentally, through in vitro tests, which we perform following the protocols set out by the OECD, and using the guidelines and recommendations available for nanomaterials and microplastics.
What type of toxicological studies do we perform?
In the case of human health risks, we work with different human origin cell cultures that enable us to study possible effects on cell functions or genetic material, as well as other effects related to irritation or inflammation problems, depending on the exposure pathway into the body.
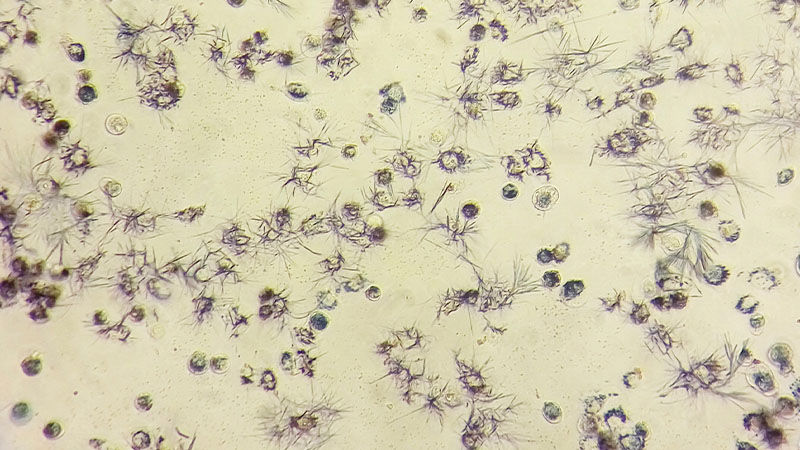
In the case of environmental risks, we study the toxic effects that these materials can have on different environmental matrices using bioassays, based on OECD and EPA protocols. We use small organisms (microinvertebrates, algae and protozoa) and plants, which are considered to be bioindicators of different matrices, to assess the potential toxic effects on the growth, mobility and reproduction of these organisms.
Tailor-made assessments and predictive models
At ITENE, we devise smart testing strategies and (eco)toxicological screening methods to assess the risks posed by exposure to chemical agents, nanomaterials and nanoproducts in industry, as well as the impact of micro- and nanoplastics on the environment. This includes drafting biosafety reports and plans, identifying active ingredients, conducting toxicity studies and toxicological profiling, and supporting risk assessment.
Our services focus on the following areas:
-
We analyse the toxicity of nanomaterials, micro- and nanoplastics, chemical additives, and mixtures. We perform toxicity and ecotoxicity studies, including:
- Assessment of cytotoxicity and genotoxicity according to the exposure pathway (inhalation, oral, or dermal).
- Assessment of dermal and ocular sensitisation and irritation.
- Biomarker analysis: generation of reactive oxygen species (ROS) and inflammatory factors.
- Acute toxicity tests on algae and micro-invertebrates through bioassays.
-
We develop, deploy and validate chemoinformatic methods such as read-across and quantitative structure-activity relationship (QSAR) mathematical models to predict the physicochemical, biological and environmental properties of chemical agents and nanomaterials. We have new software tools to draw up Safety Data Sheets and implement the REACH Regulation. Find out more about the other services we provide to support compliance with the REACH Regulation.
-
We offer consultancy services on compliance with the REACH Regulation for nanomaterials. Given the complexity of these materials, there are no standardised procedures for assessing the risks associated with them.
Benefits of toxicological studies and the use of predictive modelling
The combination of the results from toxicological studies and the application of predictive modelling is key to reinforcing safety in the use of nanomaterials and new chemical agents, and to understanding the risks they entail in the initial stages of product development. The availability of (eco)toxicological data in the early stages of new product development facilitates decision-making and the avoidance of options that could pose a risk to people’s health and the environment. Benefits include:
- Prevention and monitoring of human health risks.
- Analysis of potential toxicity in the absence of testing protocols and existing technological tools.
-
How can I help you?
Carlos Fito
Safety and Environmental Monitoring Technologies Manager